 
These Philips Trilogy 100 and Trilogy 2000 ventilators were a part of this recall in June 2021 for issues with the polyester-based polyurethane (PE-PUR) sound abatement foam breakdown. The FDA issued a safety communication to provide additional information to patients, caregivers, and health care providers about two recent issues in certain reworked Philips Trilogy 100 and Trilogy 200 ventilators. CPAP machines are often prescribed to people with obstructive sleep apnea to keep their airways open during sleep. A CPAP machine keeps your airway open by providing a stream of air at a continuous pressure through a mask.These devices are also prescribed to people with obstructive sleep apnea to keep their airways open during sleep. BiPAP machines use a higher pressure when you breathe in and lower pressure when you breathe out. A BiPAP machine pumps air under varying pressure into the airway of the lungs.A continuous ventilator mechanically controls or helps patients' breathing while delivering a set amount of oxygen.Three types of devices have been recalled: Philips also recalled certain Trilogy Evo ventilators distributed from Apto with specific serial numbers. 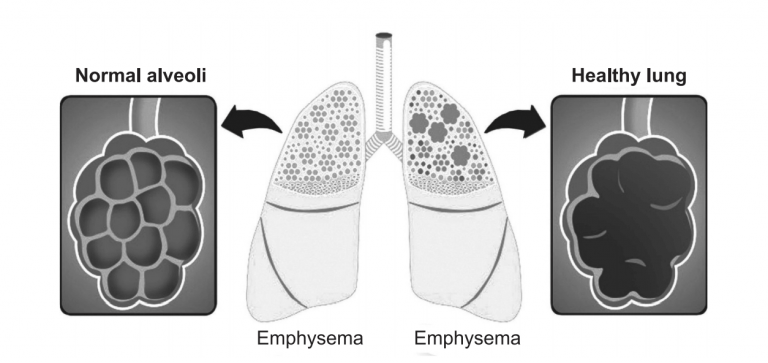
Garbin Plus, Aeris, LifeVent (ventilator).Philips recalled the following devices made between 2009 and April 26, 2021: If you use one of these recalled devices, follow the recommendations listed below. The PE-PUR foam issue may result from exposure to hot and humid conditions and may be exacerbated by the use of ozone cleaners or other cleaning methods not recommended by the manufacturer. These issues could potentially result in serious injury and require medical intervention to prevent permanent injury. If this occurs, black pieces of foam, or certain chemicals that are not visible, could be breathed in or swallowed by the person using the device. The polyester-based polyurethane (PE-PUR) foam used in these devices to reduce sound and vibration can break down. In June 2021, Philips recalled certain ventilators, BiPAP machines, and CPAP machines (see table below) because of potential health risks. FAQs on Philips Respironics Ventilator, BiPAP Machine, and CPAP Machine Recalls.Potential Health Risks from the PE-PUR Foam.Repaired and Replaced BiPAP or CPAP Machines & Recommendations.Recalled BiPAP or CPAP Machines & Recommendations.At this time, the FDA is not changing the recommendations from the previous June 2021 safety communication. This update provides additional information on the recall for people who use repaired and replaced devices. Food and Drug Administration (FDA) is updating the June 2021 safety communication about the Philips Respironics (Philips) recalled ventilators, bilevel positive airway pressure (also known as Bilevel PAP, BiPAP, or BPAP) machines, and continuous positive airway pressure (CPAP) machines. The pleural fluid LDH is less than 2/3 of the upper reference limitĪlthough these criteria have been re-evaluated there is no clear cut case for using anything other than Light’s criteria.UPDATE - June 2, 2023: This safety communication was updated to provide information about medical device reports (MDRs) the FDA received from January 1, 2023, to March 31, 2023, that are reportedly associated with the breakdown or suspected breakdown of the polyester-based polyurethane (PE-PUR) foam used in the Philips Respironics ventilators, BiPAP machines, and CPAP machines included in the recall announced in June 2021. The ratio of pleural fluid LDH to plasma LDH less than 0.6 The ratio of pleural fluid protein to serum protein is less than 0.5 The sample cannot be analysed if it is not suitable for analysis on the blood gas analyser Pleural fluid pH if a specimen in a blood gas syringe is available.Pleural fluid protein, glucose and LDH and serum protein, glucose and LDH if available. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
